

工作时间 :
周一~~周五
9:00 -18:00
在非工作时间,您可以通过邮件订购产品,订购时请写明详细联系方式,谢谢支持!
销售:18321282235
技术:021-60514606
传真:021-37680378


顾经理微信 扫一扫,关注我们
Recombinant Human Erythropoietin alpha, Liquid
Synonyms Epoetin
Accession P01588
GeneID 2056
Source CHO
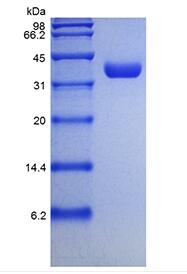
Molecular Weight Mature human EPO, containing 166 amino acid residues, has a predicted molecular mass of approximately 21 kDa. As a result of glycosylation, the recombinant protein migrates with an apparent molecular mass of 36-40 kDa in SDS-PAGE.
Quantity 500IU/2000IU/150000IU
AA Sequence APPRLICDSR VLERYLLEAK EAENITTGCA EHCSLNENIT VPDTKVNFYA WKRMEVGQQA VEVWQGLALL SEAVLRGQAL LVNSSQPWEP LQLHVDKAVS GLRSLTTLLR ALGAQKEAIS PPDAASAAPL RTITADTFRK LFRVYSNFLR GKLKLYTGEA CRTGDR
Purity > 98 % by SDS-PAGE and HPLC analyses.
Concentration See the label.
Biological Activity Fully biologically active when compared to standard. The Specific Activity was measured by the stimulation of reticulocyte production in normocyth-aemic mice and was found to be no less than 1.5 × 105 IU/mg.
Physical Appearance Clear colorless liquid.
Formulation Sterile filtered liquid in sodium citrate buffer (1 liter of ddH2O containing 5.9 g of sodium citrate, 5.8 g of sodium chloride and 0.06 g of citric acid).
Endotoxin Less than 0.01 EU/μg of rHuEPO-α as determined by LAL method.
Stability & Storage Do not freeze.
- 6 months from date of receipt, 2 to 8 °C as supplied.
- 3 months, 2 to 8 °C under sterile conditions after opening.
Usage This material is offered by Shanghai PrimeGene Bio-Tech for research, laboratory or further evaluation purposes. NOT FOR HUMAN USE.
Reference 1. Hanicki Z. 1988. Pol Arch Med Wewn, 80: 290-3.
2. Haniu M, Narhi LO, Arakawa T, et al. 1993. Protein Sci, 2: 1441-51.
3. Baranowska-Daca EandKsiazek A. 1994. Ann Univ Mariae Curie Sklodowska Med, 48 Suppl 3: 51-60.
4. Chow KM, Szeto CC, Li PK. 2003. Am J Kidney Dis, 41: 266-7; author reply 7.
5. Maurer MH, Schabitz WR, Schneider A. 2008. Curr Med Chem, 15: 1407-11.
Background Erythropoietin (EPO), a glycoprotein produced primarily by the kidney, is the principal factor that regulates erythropoiesis by stimulating the proliferation and differentiation of erythroid progenitor cells. The production of EPO by kidney cells is increased in response to hypoxia or anemia. Recombinant EPO has been approved for the treatment of anemia associated with chronic renal failure as well as for anemia of AZT treated AIDS patients. The cDNAs for EPO have been cloned from human, murine, canine, etc. The mature proteins from the various species are highly conserved, exhibiting greater than 80% sequence identity at the amino acid level. Human EPO cDNA encodes a 193 amino acid residue precursor protein that is processed to yield a 165 amino acid residue mature protein. EPO contains one O-linked and three N-linked glycosylation sites. Glycosylation of EPO is required for EPO biological activities in vivo. EPO exhibits structural as well as amino sequence identity to the amino terminal 153 amino acid region of thrombopoietin.
部门 |
姓名 | 手机 | |||
| 销售部 | 顾先生 | 1916510334@qq.com | 18321282235 | 1916510334 | |
| 技术部 | 技术支持 | 1781364813@qq.com | 13816899465 | 1781364813 |
全国免费电话:18321282235
销售: 18321282235
86-21-60514606
技术: 13816899465
传真: 021-37680378
- Recombinant Human Epidermal Growth Factor GMP...
- Recombinant Human Galectin-1...
- Recombinant Human Interleukin-36 alpha, 158a.a....
- Recombinant Rat Beta-defensin 1...
- Recombinant Human Cyclin-Dependent Kinase Inhibitor 2A, Isoform 1-TAT...
- Recombinant Human Parathyroid Hormone 7-84, 15N Stable Isotope Labeled...
- Recombinant Human Lymphocyte Activation Gene 1 Protein/CCL4L1...
- Recombinant Murine Macrophage Inflammatory Protein-3 alpha/CCL20...
- Recombinant Rat Beta-defensin 4...
- Recombinant Porcine Interleukin-1 beta...








